Breaking Through the Blood-Brain Barrier

Novel approaches aim to overcome one of the major hurdles in developing new treatments for diseases including Alzheimer’s and brain cancer.
- Most therapies struggle to access the brain, which is guarded by tightly-packed cells.
A new wave of innovation is taking aim at one of medicine’s most stubborn obstacles: the blood-brain barrier.
Diseases of the brain are notoriously difficult to treat. One problem? It is hard for most drugs to get into the brain in the first place. A protective shield of tightly-packed cells prevents pathogens and other harmful substances from reaching this vital organ. But those strong defenses have a downside: They largely keep out many therapies that could treat devastating diseases, including Alzheimer’s.
“The blood-brain barrier has been one of the major challenges that has held back the field for a very long time,” says Cillian King, a partner at EQT Life Sciences.
Scientists, however, are developing new ways to get past this biological roadblock, from smuggling treatments across it to temporarily creating gaps.
“For many years, the blood-brain barrier was sort of a nihilistic topic for why we fail,” says Adam Sonabend, a professor of neurosurgery at Northwestern University. “Now there's an interesting emergence of technologies that let investigators and doctors think about the possibility of crossing the blood-brain barrier as something to be considered in the development of a new therapy.”
Disrupting the gatekeeper
A small number of drugs can pass into the brain unaided. For the rest, getting into the brain typically involves either a direct injection or dosing at very high levels, so that the quantities of the treatment are enough to have an effect.
Scientists are racing to come up with novel approaches to overcome the challenges.
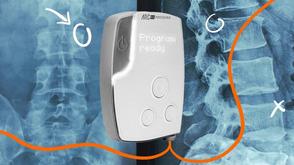
One involves temporarily creating holes in the barrier using the force released from the vibrations of tiny bubbles. These bubbles – too small to block any blood vessel – are injected into the bloodstream. A device delivers a few pulses of ultrasound to the part of the brain where treatment is needed, causing the bubbles to resonate, like an opera singer making a wine glass sing when they hit a particular note.
The force of the pulsating bubbles opens up gaps at the location exposed to the ultrasound waves, granting access to medicine circulating in the bloodstream. The ultrasound procedure is typically carried out some time after the drug is given to ensure that it has reached peak concentration in the blood at the time the barrier is opened.
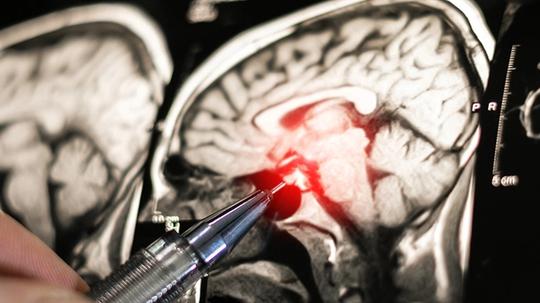
Small studies have shown that the procedure boosts the ability of powerful chemotherapy drugs to cross the blood-brain barrier, leading to larger studies in brain cancer. The approach is also being tried in Alzheimer’s.
Sonabend, the Northwestern professor, notes that this approach could have the further benefit of maintaining a higher concentration of drug in the brain than the bloodstream for a period of time because the drug can get trapped after the barrier closes.
“It’s not only the concentrations of drugs that are elevated, but they’re elevated for longer,” he says. “So the exposure of a drug to, in this case, cancer cells is greatly increased.”
Temporarily opening the blood-brain barrier also creates the possibility of tracking the progress of treatment of brain cancer, allowing molecular clues about the status of a tumor, usually confined to the brain, to flow into the bloodstream.
Hitching a ride into the brain
Another approach is allowing drugs to hitch a ride on the routes used by nutrients and other substances that are routinely granted access to the brain. EQT’s King likens this to giving the drugs a molecular “key card” that gets them past the usual obstacles.
One treatment, already available in Japan and in advanced trials elsewhere, uses this strategy for Hunter syndrome, a rare inherited disease that causes progressive damage to the body and brain. By interacting with a receptor that controls the uptake of iron into the brain, the drug can be shuttled through as if it were carrying the essential mineral.
Researchers also see promise in Alzheimer’s. Studies have shown that a drug using this approach can clear the amyloid plaques that are implicated in Alzheimer’s faster than existing treatments. That drug has now moved into large-scale human trials. Researchers are also exploring other receptors found on the blood-brain barrier as potential “shuttles” into the brain to treat Alzheimer’s and other neurological conditions like Parkinson’s.
Tipping point
“This is a great leap forward from where we are,” King says, “but it’s not perfect.” Drugs exploiting iron receptors can cause anemia in some patients and newer technologies that make use of other receptors in the blood-brain barrier might come with fewer side effects, he adds.
These promising strategies are just the tip of the iceberg, says David Dickens, a lecturer in the Department of Pharmacology & Therapeutics at the University of Liverpool.
“We’re at a tipping point,” he says, pointing to the various drugs and approaches in clinical trials, deeper insights into the molecular make-up of the blood-brain barrier and the increasing sophistication of lab-grown imitations to run experiments on. “Those things combined make it a very exciting area to be involved in.”
ThinQ by EQT: A publication where private markets meet open minds. Join the conversation – [email protected]
On the topic ofHealthcare
Exclusive News and Insights Every Week
Sign up to subscribe to the EQT newsletter.